ABOUT GENOVIOR
Genovior is a dedicated CDMO focus on process development, scale up, formulation development, and productivity optimization under PIC/S GMP (cGMP, EU GMP) for our customers worldwide. Our one-stop services conveniently provide effective and efficient solution and our non-competition policy provides maximum security and protection in IP for our clients.
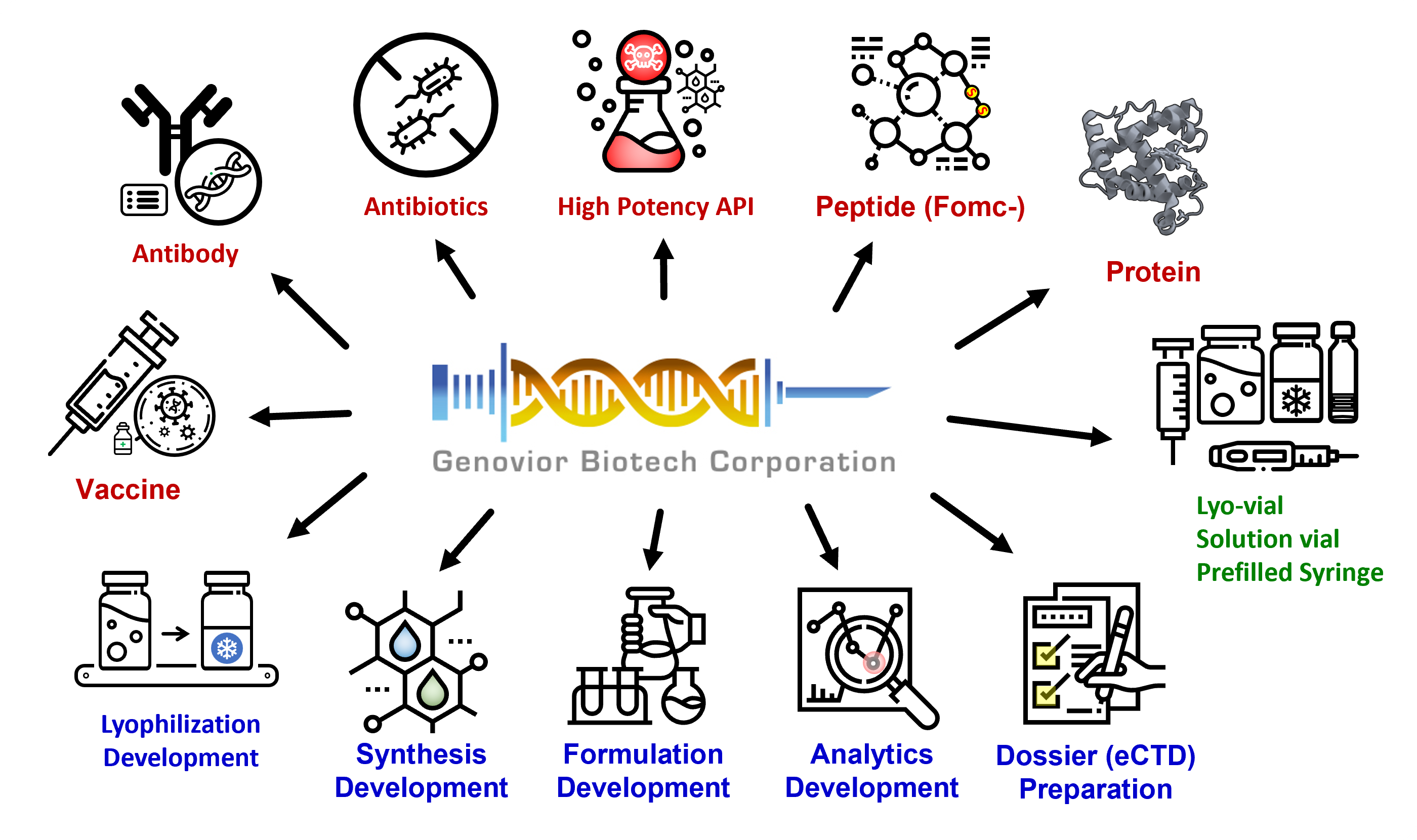
Genovior Biotech Corp. is a vertically integrated contract development and manufacture company, providing following services:
(a) process development of peptide or protein biologics from microbial or human recombinant fermentation processes
(b) formulation development of biological injectables
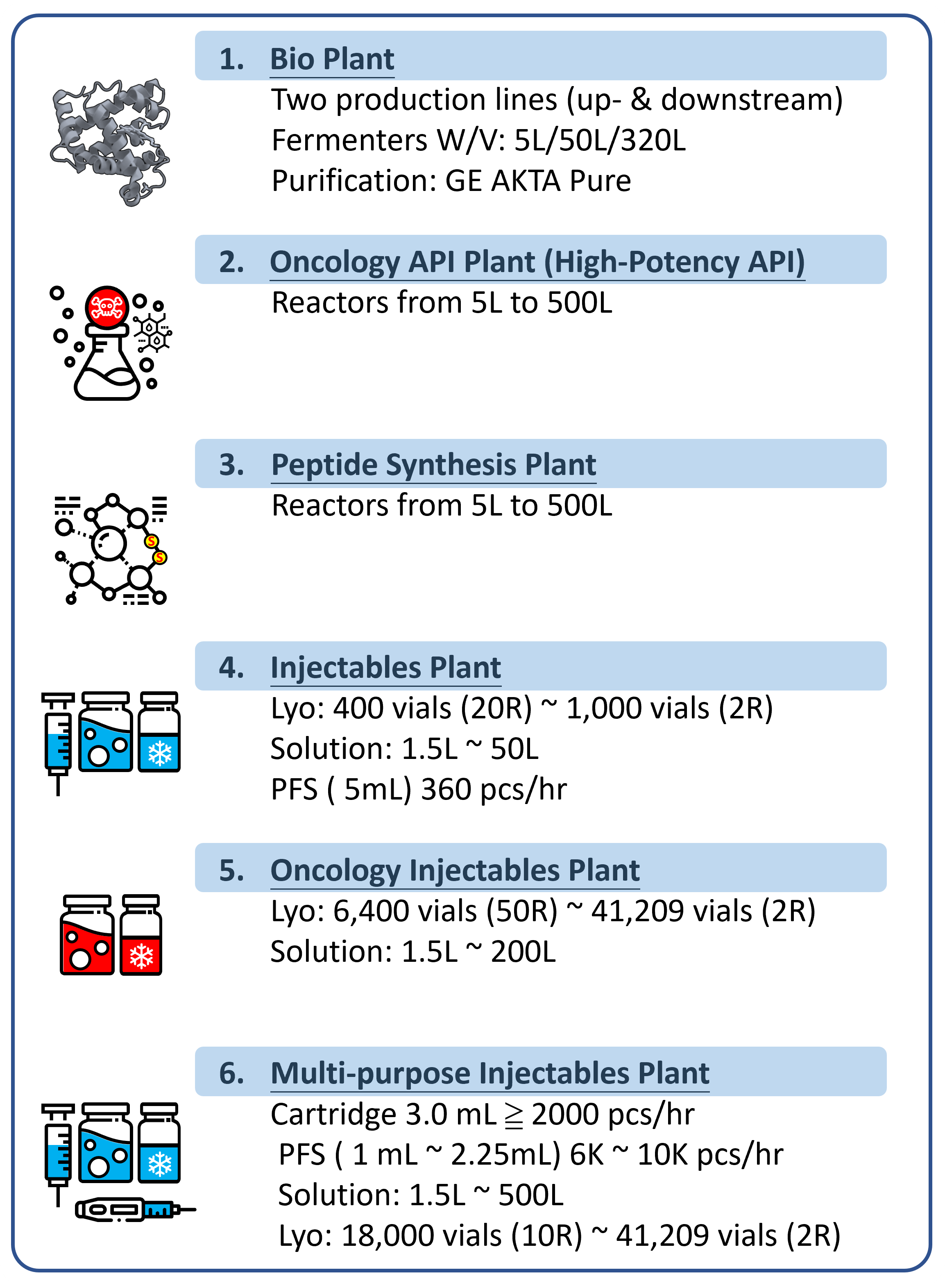
(c) manufacturing capacities of biologics, lyophilized injectable, and prefilled syringe/cartridge for preclinical, clinical, and commercial uses
SERVICES
-
 SynthesisGenovior provides professional services in (a) process development, (b) scale up, and (c) small scale production...
SynthesisGenovior provides professional services in (a) process development, (b) scale up, and (c) small scale production... -
 Protein Process DevelopmentGenovior is capably of developing protein process for the products, either new or generic biologics, initiated b...
Protein Process DevelopmentGenovior is capably of developing protein process for the products, either new or generic biologics, initiated b... -
 Formulation DevelopmentGenovior can formulate a long-acting release delivery systems of PEGylation technology, microsphere, nanoparticl...
Formulation DevelopmentGenovior can formulate a long-acting release delivery systems of PEGylation technology, microsphere, nanoparticl... -
 Analytical DevelopmentGenovior provides services in analytical method development, qualification, and validation ...
Analytical DevelopmentGenovior provides services in analytical method development, qualification, and validation ...